From: https://jamanetwork.com/journals/jama/fullarticle/2777059
JAMA Insights
February 26, 2021
SARS-CoV-2 Vaccines
C. Buddy Creech, MD, MPH1; Shannon C. Walker, MD1; Robert J. Samuels, MBChB1
Author Affiliations Article Information
JAMA. Published
online February 26, 2021.
doi:10.1001/jama.2021.3199
Audio Clinical Review
(32:50)
Coronavirus Vaccines—An Overview
0:00 / 0:00
Shortly after SARS-CoV emerged at the turn of the 21st century, the spike (S) protein
(particularly in its prefusion [native]
conformation) was identified as the immunodominant antigen of the virus.1 [The
presence of antigens in the body normally triggers an immune response.] Evaluation of patients with SARS-CoV-2 revealed that
binding and neutralizing antibodies primarily target the receptor-binding
domain of the S1 subunit.2 Once this putative vaccine target
was identified, the next challenge was how to best generate an effective immune response to
SARS-CoV-2. The characteristics of this response would include
1. production of neutralizing antibodies,
2. generation of a T-cell response, and
3. avoidance of immune-enhanced disease
(vaccine-induced response that led to paradoxically increased disease severity on viral
challenge).3
Several vaccine designs were evaluated by
different groups during the development of a SARS-CoV-2 vaccine. The SARS-CoV-2
vaccines currently authorized for use, and others that have late-stage clinical
data available, are summarized in the Table.
Table. SARS-CoV-2 Vaccines
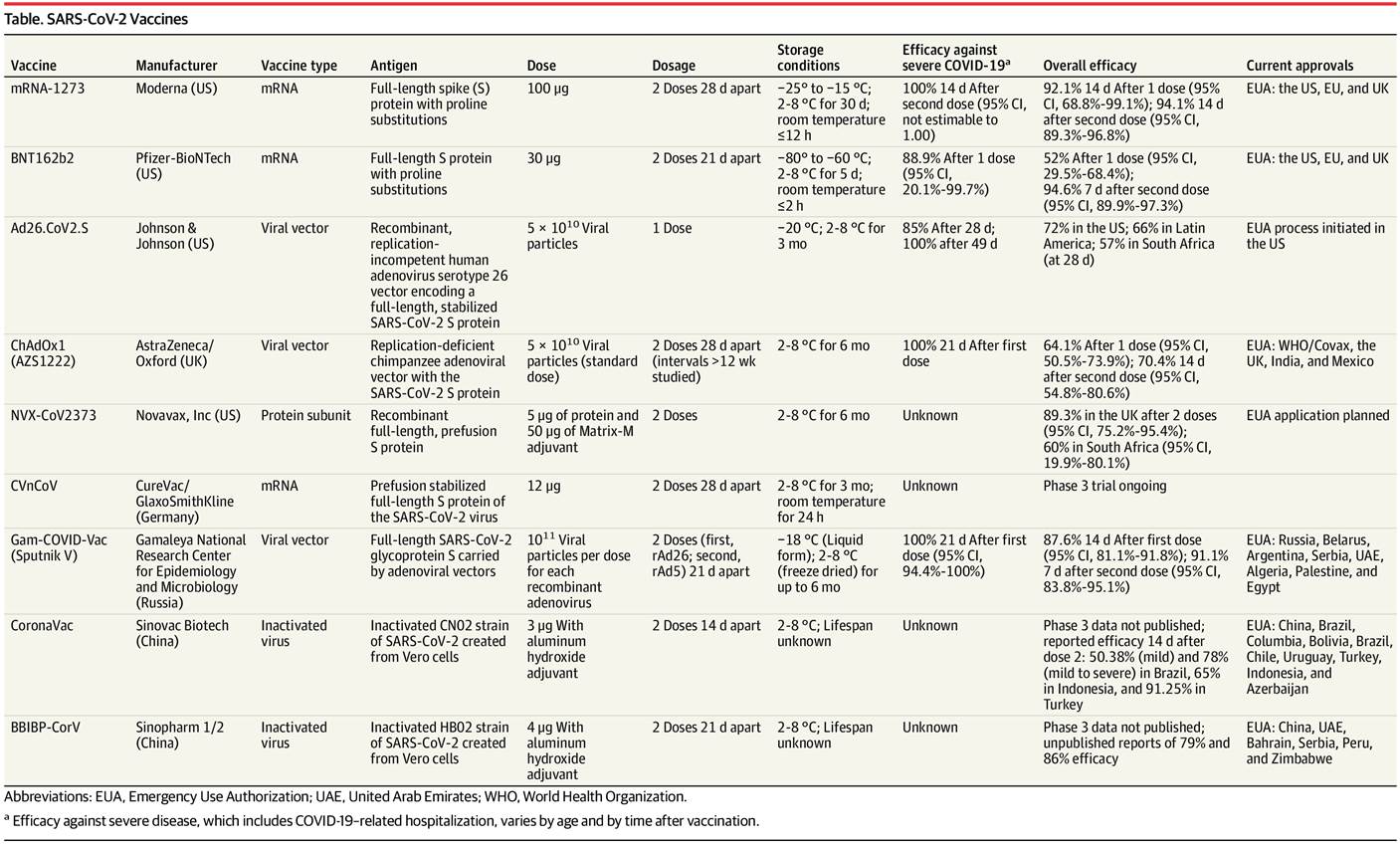
Inactivated and Protein Subunit Vaccines
One approach for vaccine development is
creation of inactivated vaccines derived from virus grown in culture and then
chemically inactivated, which may deliver stably expressed, conformationally
native antigenic epitopes. Sinopharm
and Sinovac are
among the manufacturers farthest along in development of this type of vaccine,
which have been evaluated by phase 3 trials that have attained international
authorizations for use.
Another approach to vaccine development is
delivery of the S protein as a recombinant protein subunit within one of
several cell-based systems that support protein expression. This approach can
protect immunized animals in vivo but has the theoretic risk of generating a
polarized (TH2 over TH1) immune response that can be
overcome, depending on the adjuvant used.4 Novavax, using the saponin-based Matrix-M
adjuvant, recently reported on its late-phase clinical trials in the UK,
demonstrating vaccine efficacy against COVID-19 of 89%.5 More than 60% of vaccines
currently in development use a protein subunit approach, although none are
authorized for use.
Viral Vector Vaccines [A vaccine is a biological preparation that provides active acquired immunity to a
particular infectious disease.[1] ]
Viral vector vaccines use replication-deficient viruses engineered to express the genetic
sequence of the antigen of interest in host cells. Replication-incompetent
adenoviruses have been developed for HIV, tuberculosis, malaria, and Ebola
virus.6 This vaccination approach has had
variable success, often limited by preexisting immunity to the adenovirus
vector.7 Using adenoviruses that have
minimal preexisting immunity in the US and Europe, 2 vaccines have shown early
promise: adenovirus serotype 26 vector vaccine (Ad26.CoV2.S; Johnson & Johnson) and chimpanzee
adenovirus vector vaccine (ChAdOx;
AstraZeneca). Both appear efficacious in preventing
COVID-19–related hospitalization and death, but have varying efficacy in preventing clinical disease,
particularly disease caused by the novel SARS-CoV-2 variants.
mRNA Vaccines
[A vaccine is a biological preparation
that provides active acquired immunity to a
particular infectious disease.[1] ]
New advancements harnessing mRNA for vaccine
delivery have the potential to greatly
improve vaccine development for many pathogens. In these vaccines, lipid
nanoparticles are used to protect the prefusion-stabilized S protein–encoding mRNA en route
to the intracellular space. The host [human] uses the mRNA to make the target
protein (S protein
in this case), which induces a coordinated immune response. Pfizer-BioNTech and Moderna have developed
mRNA-based vaccines that demonstrate more than 90% efficacy against SARS-CoV-2
clinical disease in clinical trials. This high vaccine efficacy is associated
with very few adverse events, although local and systemic reactogenicity to [the] vaccine are common. There are many advantages to this approach,
including speed of vaccine manufacturing (weeks) and ability to generate a TH1
and TH2 [?] response. Studies are underway or planned to
assess the efficacy of currently authorized vaccines in children and against
common SARS-CoV-2 variants, and to assess whether repeat vaccinations
containing mRNA coding for the variants can be effective.
Vaccines Are Available, What Next?
Once vaccines became available, barriers to
administration included insufficient initial supply, vaccine delivery
inefficiencies, and widespread vaccine hesitancy. These barriers limited the
ability to vaccinate enough of the population to reach some measure of
population immunity. Outside of the US, low- and middle-income countries have
struggled to obtain even a minimum number of vaccine doses.
The slower-than-hoped-for vaccine rollout
raises 2 important public health questions.
The first is whether it is preferable to
ensure maximal coverage by vaccinating as many people as possible with 1 dose
(of the 2-dose vaccines) or to ensure maximal protection by strategically
reserving doses to be used for the second dose. Based on the US Food and Drug
Administration briefing materials submitted for Emergency Use Authorization,
the Moderna vaccine is upwards of 80% efficacious 2 weeks after the first dose
and the Pfizer-BioNTech vaccine is at least greater than 50% efficacious after
the first dose.
Second, optimizing vaccination strategies in
people previously infected with SARS-CoV-2 offers another opportunity for dose
sparing. A single dose of an mRNA vaccine might quickly and robustly boost a
previously primed immune response following natural disease.
Vaccination is the most important strategy to
end the pandemic. However, emergence of multiple SARS-CoV-2 variants with
reduced susceptibility to disease- and vaccine-induced immunity threatens
progress. Despite these ongoing threats, the efficacy of SARS-CoV-2 vaccines
provides a real measure of hope for 2021.
Article Information
Corresponding Author: C. Buddy Creech MD, MPH, Division of
Infectious Diseases, Vanderbilt University School of Medicine, D-7235 MCN,
Nashville, TN 37232-2581 (buddy.creech@vanderbilt.edu).
Published Online: February 26, 2021. doi:10.1001/jama.2021.3199
Conflict of Interest Disclosures: Dr Creech reported receiving personal fees from
Altimmune for vaccine development and from Horizon for care of children with
chronic granulomatous disease; grants from Merck for Clostridioides
difficile treatment and from GlaxoSmithKline for Staphylococcus
aureus vaccine development; and personal fees from Premier Healthcare
for vaccine education, Astellas for serving on a vaccine study data and safety
monitoring board, Karius Diagnostics, and Vir Biotechnology for monoclonal
antibody development outside the submitted work. No other disclosures were
reported [from any of the
three authors of this report].
References
1.
Du L, He Y, Zhou Y, et al. The spike protein of SARS-CoV—a target for
vaccine and therapeutic development. Nat Rev
Microbiol. 2009;7(3):226-236.PubMedGoogle ScholarCrossref
2.
Premkumar L, Segovia-Chumbez
B, Jadi R, et al.
The receptor binding domain of the viral spike protein is an
immunodominant and highly specific target of antibodies in SARS-CoV-2
patients. Sci Immunol.
2020;5(48):eabc8413.PubMedGoogle Scholar
3.
Tseng CT, Sbrana E, Iwata-Yoshikawa
N, et al. Immunization with SARS
coronavirus vaccines leads to pulmonary immunopathology on challenge with the
SARS virus. PLoS One. 2012;7(4):e35421.PubMedGoogle Scholar
4.
Zimmermann P, Curtis N. Factors that influence the immune response to
vaccination. Clin Microbiol Rev.
2019;32(2):e00084-18.PubMedGoogle Scholar
5.
Novavax COVID-19 vaccine demonstrates 89.3% efficacy in UK phase
3 trial. News release. January 28, 2021. Accessed February 21, 2021. https://ir.novavax.com/news-releases/news-release-details/novavax-covid-19-vaccine-demonstrates-893-efficacy-uk-phase-3
6.
Tatsis N, Ertl HC. Adenoviruses
as vaccine vectors. Mol Ther.
2004;10(4):616-629.PubMedGoogle ScholarCrossref
7.
Zak DE, Andersen-Nissen E, Peterson ER, et al.
Merck Ad5/HIV induces broad innate immune activation that predicts CD8+ T-cell
responses but is attenuated by preexisting Ad5 immunity. Proc Natl Acad Sci U S A.
2012;109(50):e3503-e3512.PubMedGoogle ScholarCrossref
Comment